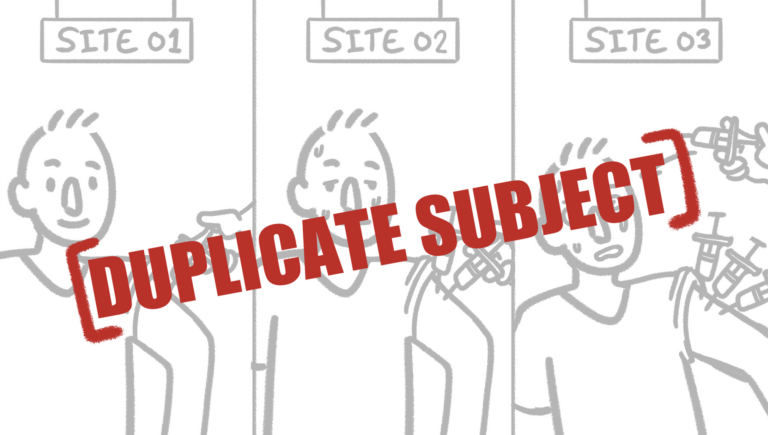
Beyond Their Lane: How Verified Clinical Trials Detects Cross-Therapeutic Duplicate Subjects In Clinical Trials
In the clinical research world, it’s easy to assume that participants stay in their lane—only enrolling in studies within the same therapeutic area. The reality is far different. Research subjects often cross over between very different indications, creating hidden risks for trial integrity, data quality, and participant safety. These situations aren’t limited to professional subjects …