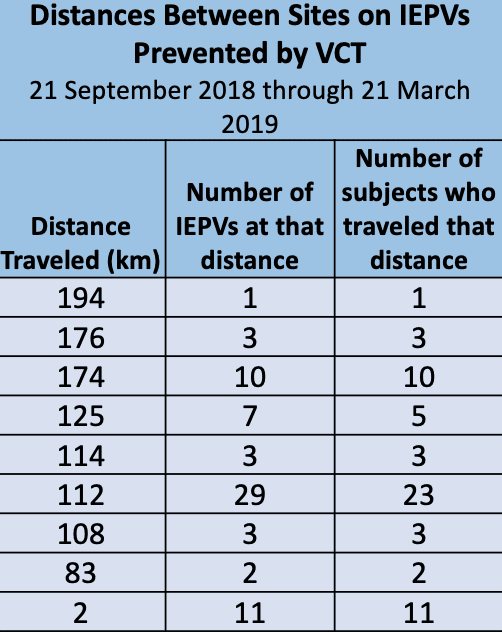
Verified Clinical Trials (VCT) has been well adopted in many research sites and CROs in several European countries. Data again proves that research volunteers will travel significant distances to dual enroll in multiple clinical trials contemporaneously. There is a similar experience in the United States whereby duplicate subjects in clinical trials will travel thousands of miles at times. As the network continues to expand and more data is compiled, we expect to see more of this phenomenon.
A global subject registry such as Verified Clinical Trials (VCT) will detect and prevent duplicate subjects in clinical trials and professional research subjects. This will improve research subject safety and data quality across all phases of clinical trials research.