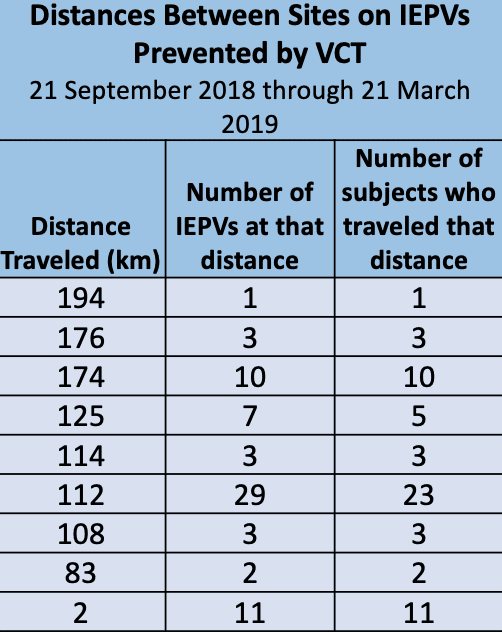
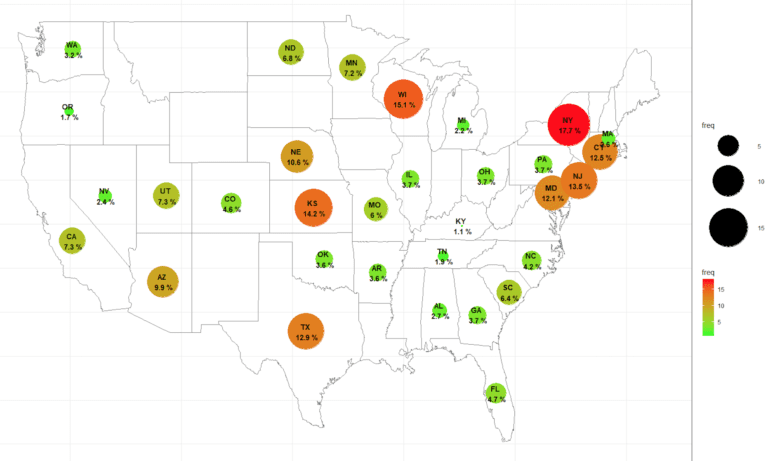
Strategies To Proactively Detect And Prevent Professional Research Subjects With Verified Clinical Trials Research Subject Database Registry
Clinical trials around the world face challenges from professional research subjects or dual enrollment in clinical trials. One group of subjects that have high rates of participation in clinical research are subjects who enroll in multiple trials for the purpose of generating income through study payments or using clinical trials as medical care. These subjects …