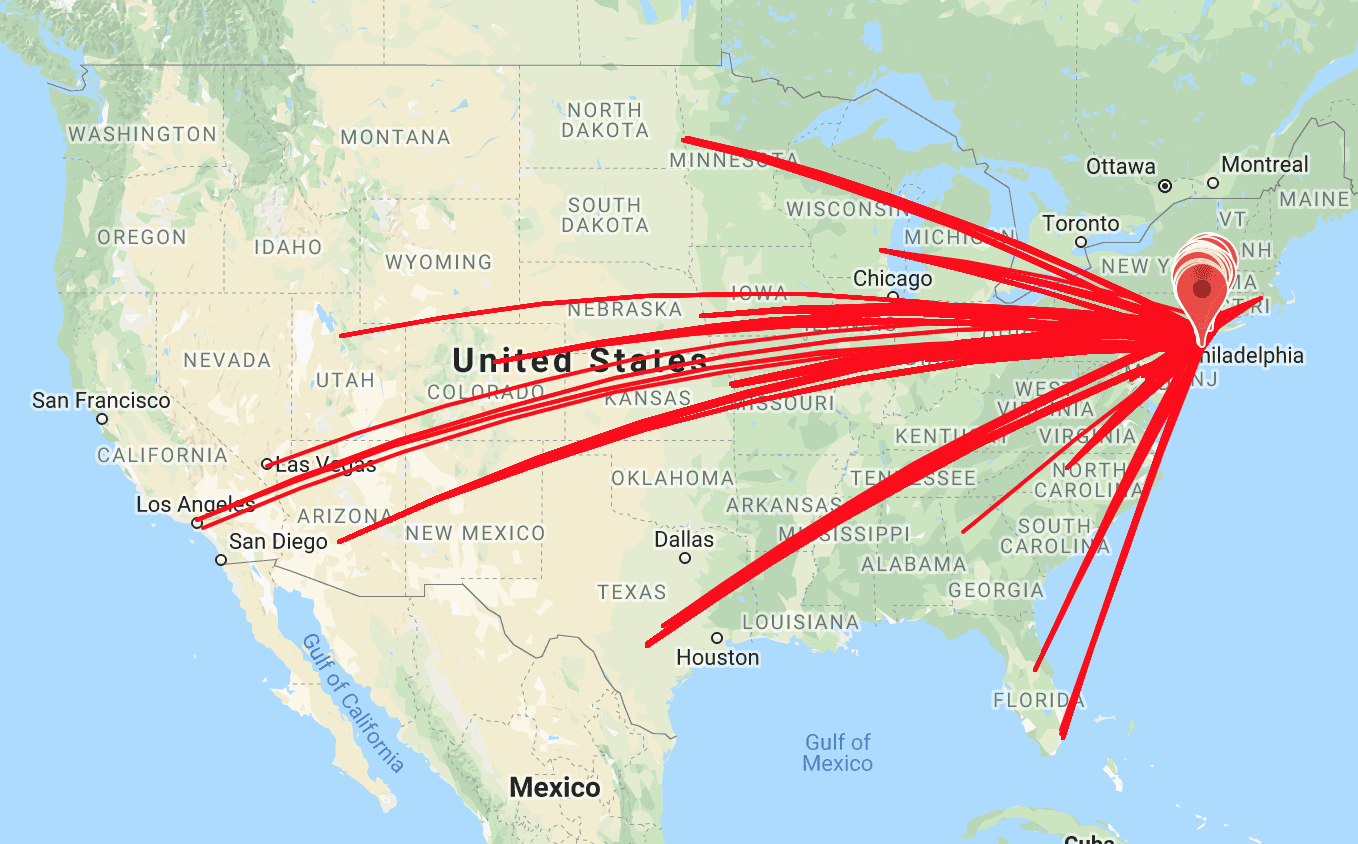
Duplicate subjects in clinical trials has attracted the attention of many clinical trial managers and study leads. To prevent duplicate subjects or professional research subjects, many sponsors have added the use of Verified Clinical Trials (VCT)
as a mandatory step during the screening process. VCT verifies a research subject’s study history at the time of screening and therefore prevents costly screenings saving the site, CRO, and sponsor time and money.
VCT has excellent data showing that research subjects will travel from California to New York, thousands of miles, to enroll contemporaneously in multiple trials.